|
Overexpression of the succinylation mimetic LDHA K155E mutant restored glycolytic metabolism and cell viability in cells in which metabolic reprogramming and cell viability were ceased due to GLTC depletion. 
Mass spectrometry revealed that GLTC, as a binding partner of LDHA, promotes the succinylation of LDHA at lysine 155 (K155) via competitive inhibition of the interaction between SIRT5 and LDHA, thereby promoting LDHA enzymatic activity. 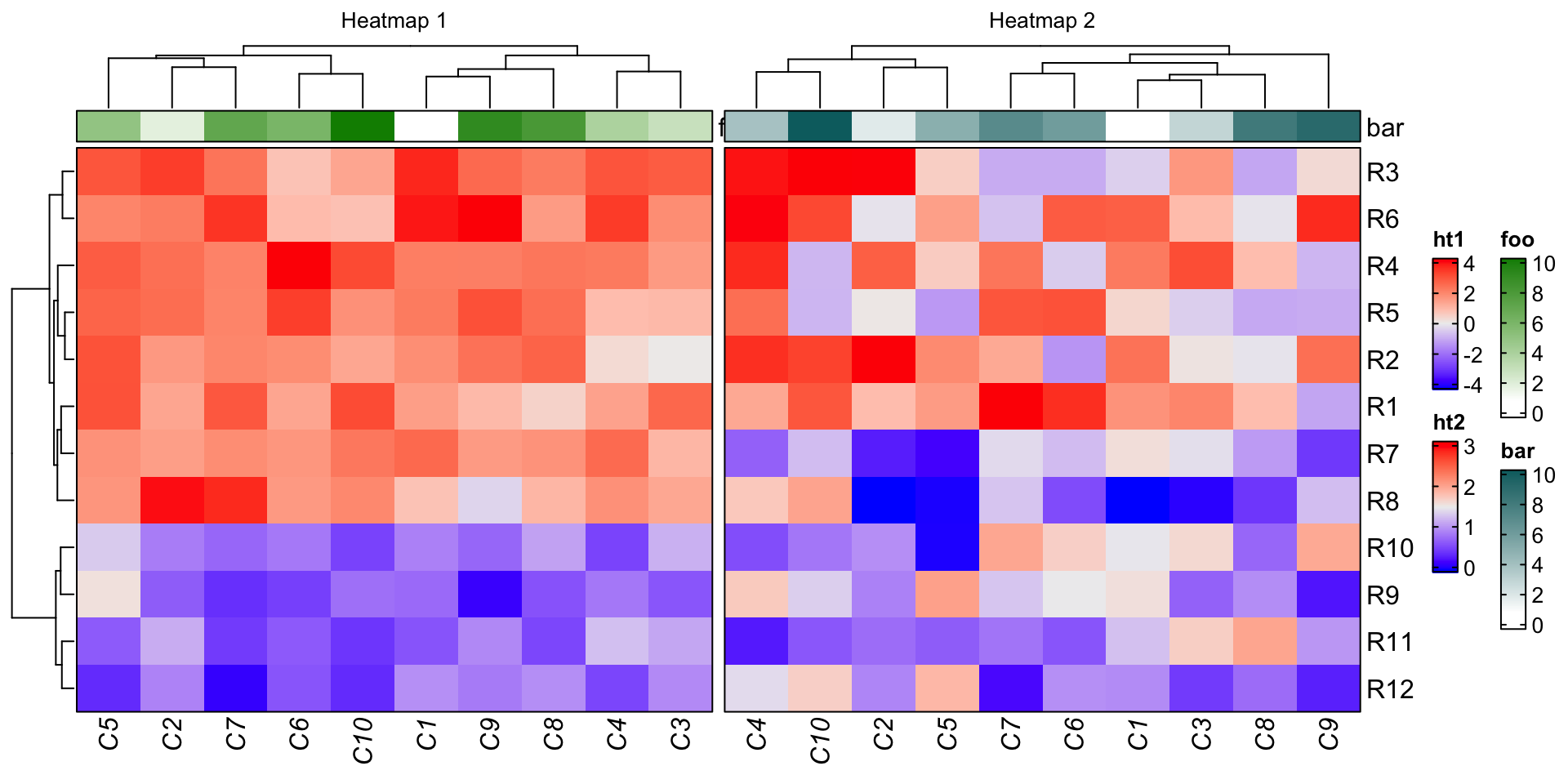
High expression of GLTC was correlated with more extensive distant metastasis, a larger tumour size, and poorer prognosis. GLTC was significantly upregulated in PTC tissues compared with nontumour thyroid tissues. Through screening, we identified an LDHA-interacting lncRNA, GLTC, which is required for the increased aerobic glycolysis and cell viability in PTC. However, whether specific lncRNAs can regulate LDHA activity during cancer progression remains unclear. Lactate dehydrogenase A (LDHA) enzymatic activity is also crucial for cancer development, including the development of papillary thyroid cancer (PTC). 
Dysregulation of long noncoding RNAs (lncRNAs) has been associated with the development and progression of many human cancers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
